Rohan Ahmed
About Me
With over 08 years of hands-on experience in the highly regulated Pharmaceuticals and third-party Testing (MNC) industry, along with the Education and Analytical Instrument Sales sectors in Bangladesh, I have developed deep expertise in Quality Assurance (QA) and Quality Control (QC).
My background includes extensive work with QA/QC Analytical Instruments, including HPLC, GC, GCMS, ICP-OES, ICP-MS, AAS, FTIR, UV, FL, KF, Potentiometers, Dissolution Testers, DT, Polarimeters, MP/BP Meters, RI Meters, and more.
Beyond instrumentation, I have played key roles in Quality Management Systems (QMS), strengthening my expertise in Technical Documentation (especially Preparation and Updating of SOPs, Specifications, STPs; Change Control, OOS, OOT, Incident, Deviation), Data Review, Electronic Signature, Data Integrity (DI), Data Backup/Restore on Server, Audit Trails, and Standard QA/QC Procedures—all critical for maintaining compliance and operational excellence in the pharmaceutical sector.
Additionally, I have had the privilege of handling recruitments for both QA & QC Department, and conduct training on SOPs.
On top of that, you’ll find me highly skilled in computer software applications, with professional proficiency in Graphic Design and Microsoft Productivity Tools (Photoshop, Illustrator, Excel, Word, Outlook, and PowerPoint). My ability to merge technical expertise with digital solutions allows me to contribute effectively to process optimization and documentation efficiency.
Career Goal
To LEAD a highly skilled team of a renowned organization with INTEGRITY, especially fields involving QMS (QA/QC), International Regulation, Auditing/Compliance, bringing business for the Organization and keeping the team SATISFIED through demonstrating 05 major skills: Effective COMMUNICATION, Efficient NEGOTIATION, Strategic LEADERSHIP, Target-oriented PLANNING, and Appropriate VALUATION of Skills & Expertise.
Experience
Dhaka, Bangladesh
Role Tenure: October 01, 2025 to Present
Green Trade Point, 7, Mohakhali C/A, Dhaka 1212, Bangladesh
Role Tenure: February 01, 2025 to September 30, 2025
Diabari Permanent Campus, Uttara Model Town, Uttara, Dhaka, Bangladesh
Role Tenure: August 20, 2024 to January 31, 2025
Kalachandpur, Nadda, Dhaka, Bangladesh
Role Tenure: May 19, 2024 to August 18, 2024
Dhaka, Bangladesh
Role Tenure: April 01, 2023 to April 30, 2024
Sonargaon, Narayanganj, Bangladesh
Role Tenure: February 01, 2023 to March 31, 2023
Kashor, Bhaluka, Mymensingh, Bangladesh
Role Tenure: January 01, 2022 to January 31, 2023
Plot 1, Section 7, Milk Vita Road, Mirpur, Dhaka 1216, Bangladesh
Role Tenure: April 01, 2019 to December 31, 2021
Intertek House, Phoenix Tower, Tejgaon I/A, Dhaka, Bangladesh
Role Tenure: August 03, 2017 to March 28, 2019
Education
QA/QC Analytical Instrumentation Expertise Experience Operating Knowledge
Software Skills Computer Literacy Digital Proficiency
LinkedIn Courses
Courses / Training
YouTube
Contact Me
Find Me Here
Extra Activities
Teaching
Volunteer Works
Volunteer Works
Interests
Indoor Gardening
Indoor gardening is an exciting hobby that can enhance your living space and improve your well-being. Cultivate a variety of plants and enjoy reduced stress levels and a healthier indoor environment. This hobby also offers an outlet for creativity, allowing you to experiment with different plants and arrangements to beautify your home.
Cooking
Cooking is a fun skill that allows you to create delicious meals and desserts. It's a great way to get creative, reduce stress, and make memories with friends and family.
Web Design
Passionate about web design: merging creativity and technology to craft visually captivating, user-friendly online experiences. 🌐💻
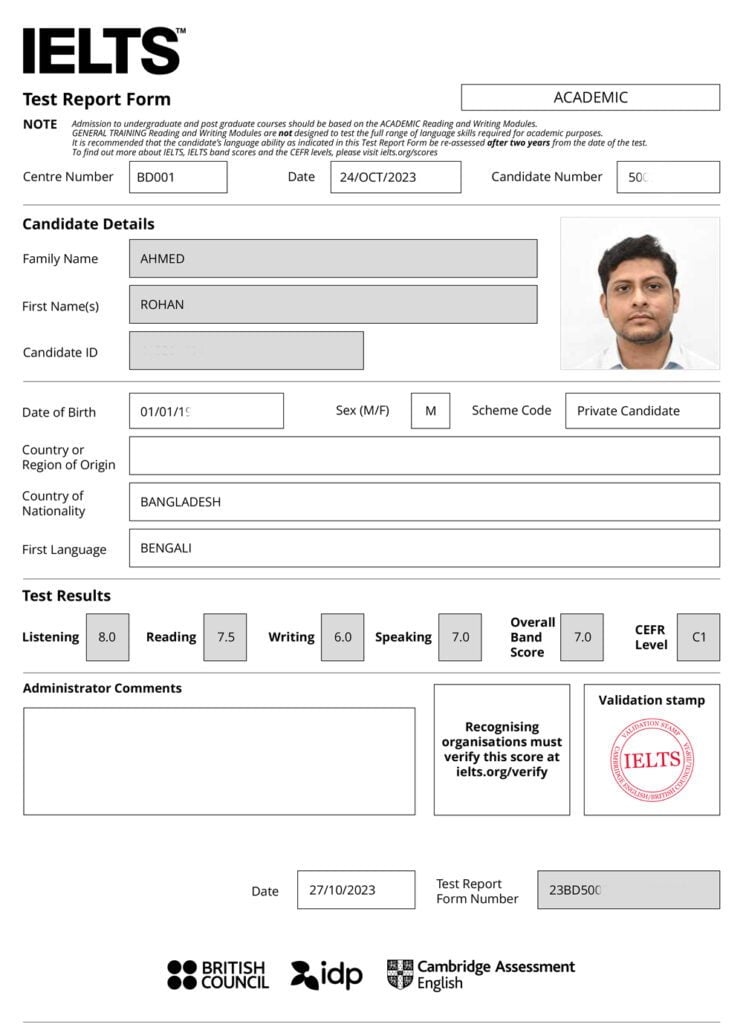
IELTS